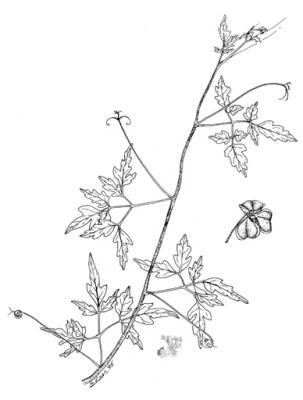
Cardiospermum halicacabum
Vernacular names
(In territories with significant traditional TRAMIL use)
Martinique:
- boné karé
Botanical description
Climbing shrub 3 (-6 m) high. Leaves alternate up to 12 cm long, compound, blade pellucid-dotted, leaflets 5 cm x 2.5 cm, ovate-elliptical, acute-tipped; inflorescence corymbose; flowers white, 4-petals, 4-6 mm long; fruit sub-globose, 3-angled much inflated capsule, 2.5-3 cm in length and width; seeds black, 4-5 mm long with white aril.
Voucher(s)
Nossin,6,HAVPM
Pharmacopoeia
Ed.2References
1 LONGUEFOSSE JL, NOSSIN E, 1990-95 Enquête TRAMIL. Association pour la valorisation des plantes médicinales de la Caraïbe AVPMC, Fort de France, Martinique.
2SOLIS P, GUPTA M, CORREA M, 1996 Estudio fitoquímico de algunas plantas TRAMIL. Informe TRAMIL. Centro de Investigaciones Farmacognósticas de la Flora Panameña CIFLORPAN, Facultad de Farmacia, Universidad de Panamá, Panamá, Panamá.
3 KUMARESAN A, 1981 Phyto-chemical investigation of the leaves of the plant Cardiospermum halicacabum Linn. Indian J Pharm Sci: D20.
4 MECKLINGER S, MESSEMER C, NIEDERLE S, 1995 Eksembehandlung mit Cardiospermum halicacabum. Zeitschrift für Phytotherapie 16(5):263-266.
5 KHAN M, ARYA M, JAVED K, KHAN M, 1990 Chemical examination of Cardiospermum halicacabum Linn. Indian Drugs 27(4):257-258.
6 SHABANA M, GENENAH A, EL ZALABANI S, ABOU EL-ELA R, YOUSIF M, 1990 Phytochemical investigation and insecticidal activity of Cardiospermum halicacabum L. Cultivated in Egypt. Bull Fac Pharm Cairo Univ 28(2):79-83.
7 KHAN M, ARYA M, JAVAED K, KHAN M, 1990 Chemical examination ofCardiospermum halicacabum Linn. Indian Drugs 27(4):257-258.
8 ABBURRA R, GUZMAN C, 1986 Phytochemical studies of the Argentine Sapindaceae. I. Oil composition in seeds of the genus Cardiospermum. An Asoc Quim Argent 74(5):571-574.
9 HOPKINS C, EWING D, CHISHOLM M, 1968 A short-chain ester from the seed oil of Cardiospermum halicacabum L. Phytochemistry 7:619-624.
10 PLOUVIER V, 1949 New investigation on quebrachitol in the Sapindaceae and Hippocastanaceae, dulcitol in the Celastraceae and sucrose in some other families. C R Acad Sci 228:1886-1888.
11 DESAI K, SETHNA S, 1954 Chemical investigation of the roots of the Indian medicinal plant Cardiospermum halicacabum. J MS Univ Baroda 111:33-39.
12HERRERA J, 1996 Validación farmacológica de plantas medicinales usadas en medicina tradicional popular en la cuenca del Caribe. Informe TRAMIL. Dep. de Farmacología, Facultad de Salud, Universidad del Valle, Cali, Colombia.
13 CHANDRA T, SADIQUE J, 1984 Antiflammatory effect of the medicinal plant Cardiospermum halicacabum Linn. In vitrostudy. Arogya 10(1):57-60.
14 DHAR ML, DHAR MM, DHAWAN BN, MEHROTRA BN, RAY C,1968 Screening of Indian plants for biological activity: Part I. Indian J Exp Biol 6:232-247.
15 GOPALAKRISHNAN C, DHANANJAYAN R, KAMESWARAN L, 1976 Studies on the pharmacological actions of Cardiospermum helicacabum. Indian J Physiol Pharmacol 20:203.
16 SADIQUE J, CHANDRA T, THENMOZHI V, ELANGO V, 1987 Biochemical modes of action of Cassia occidentalis and Cardiospermum halicacabum in inflammation. J Ethnopharmacol 19(2):201-212.
17 HORMANN H, KORTING H, 1994 Evidence for the efficacy and safety of topical herbal drugs in dermatology: Part I: anti-inflammatory agents. Phytomedicine 1(2):161-171.
18 MENG Z, SAKI Y, OSE Y, SATO T, NAGASE H, KITO H, SATO M, MIZUNO M, ONO K, NAKANE H, 1990 Antimutagenic activity by the medicinal plants in traditional Chinese medicines. Shoyakugaku Zasshi 44(3):225-229.