Vernacular names
(In territories with significant traditional TRAMIL use)
Guatemala:
- hierba de San Juan
- iyá
- jolomocox
- pericón
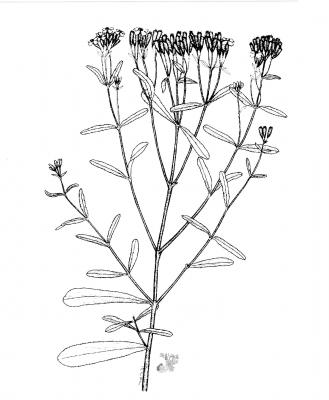
Botanical description
Annualherb, highly scented, glabrous, erect, 30-95 cm; very resinous when dried. Leaves opposite, sessile, linear or oblong-lanceolate, 5-10 mm long, obtuse or acute at the apex, finely dentate. Flowers yellow, in small terminal heads, 9-10 mm in diameter, involucre cylindrical, bracts usually 3 in number. Achenes 6-7 mm long, striate.
Voucher(s)
Girón,164,CFEH
Pharmacopoeia
Ed.2References
1 GIRON L, 1988 Encuesta TRAMIL (Costa atlántica). Centro Mesoamericano de Tecnología CEMAT, Guatemala, Guatemala.
2 HETHELYI E, DINOS B, TETENYI P, 1986 Analysis of essential oils of someTagetes species. In progress in essential oil research. Berlin, RFA: GC/MS Walter de Gruyter, p131-137.
3 ABDALA LR, 1999 Flavonoids of the aerial parts from Tagetes lucida (Asteraceae). Biochem Syst Ecol 27(7):753-754.
4 GLASBY JS, 1991 Dictionary of plants containing secondary metabolites. London, England: Taylor & Francis.
5 RODRIGUEZ E, MABRY TJ, 1977 Tagetae chemical review. In Heywood VH, Harborne JB, Turner BL, Eds. The biology and chemistry of the Compositae, Vol. II, 785-797. New York: Academic Press.
6 LAFERRIERE JE, WEBER CW, KOHLHEPP EA, 1991 Mineral composition of some traditional Mexican teas. Plant Foods Hum Nutr 41(3):277-282.
7 DUKE JA, ATCHLEY AA, 1986 Handbook of proximate analysis tables of higher plants. Boca Raton, USA: CRC Press, p156.
8 CACERES A, SARAVIA A, JAUREGUI E, AGUIRRE I, 1992 Actividad antiinflamatoria de plantas medicinales de uso popular en Guatemala (I). Informe TRAMIL. Cuadernos de la Dirección General de Investigación, Universidad de San Carlos, Guatemala, Guatemala.
9 SALGUERO IE, 1989 Estudio farmacológico de Tagetes lucida (pericón) (Tesis Mag. Sc). Facultad de Ciencias Químicas y Farmacia, Universidad San Carlos, Guatemala, Guatemala.
10 CAMBAR P, COUSIN L, SANTOS A, ALGER J, MENDOZA M, 1984 Efectos de los extractos de algunas plantas medicinales de Honduras sobre la motilidad intestinalin vitro. Tegucigalpa, Honduras: Dirección de Investigación Científica. Universidad Nacional Autónoma de Honduras.
11 CACERES A, FLETES L, AGUILAR L, RAMIREZ O, FIGUEROA L, TARACENA AM, SAMAYOA B, 1993 Plants used in Guatemala for the treatment of gastrointestinal disorders. 3. Confirmation of activity against enterobacteria of 16 plants. J Ethnopharmacol 38(1):31-38.
12 MENDEZ A, 1991 Evaluación de la actividad anti-Candida albicans in vitro de diez plantas de uso medicinal en Guatemala (Tesis Mag. Sc). Facultad de Ciencias Químicas y Farmacia, Universidad San Carlos, Guatemala, Guatemala.
13 SIDDIQUI MA, ALAM MM, 1987 Control of phytonematodes by mix-culture of Tagetes lucida. Indian J Plant Pathol 5(1):73-78.
14 SIDDIQUI MA, ALAM MM, 1989 Toxicity of different plant parts of Tagetes lucida to plant parasitic nematodes. Indian J Nematol 18(2):181-185.
15 DUKE JA, 1992 Handbook of biologically active phytochemicals and their bioactivities. Boca Raton, USA: CRC Press.
16 SARAVIA A, 1992 Estudios sobre plantas TRAMIL. Departamento de farmacología, Facultad de Ciencias Químicas y Farmacia, Universidad San Carlos, Guatemala, Guatemala. TRAMIL VI, Basse Terre, Guadeloupe, UAG/enda-caribe.
17 CHAN GFQ, Lee MM, Glushka J, Towers GHN, 1979 Photosensitizing thiophenes in Porophyllum, Tessaria and Tagetes. Phytochemistry 18(9):1566.
18 MORTON J, 1981 Atlas of medicinal plants of Middle America: Bahamas to Yucatan. Springfield, USA: Charles C. Thomas Publisher.
19 GIRON L, CACERES A, FREIRE V, ALONZO A, SALVADOR L, 1995 Folleto informativo sobre algunas plantas comúnmente utilizadas por la población Garífuna de Livingston, Guatemala, Guatemala, p41.
20 CACERES A, LOPEZ B, GONZALEZ S, BERGER I, TADA I, MAKI J, 1998 Plants used in Guatemala for the treatment of protozoal infections. I. Screening of activity to bacteria, fungi and American trypanosomes of 13 native plants. J of Ethnopharmacology 62(3):195-202.