Vernacular names
(In territories with significant traditional TRAMIL use)
Haiti:
- koray
- koway
Botanical description
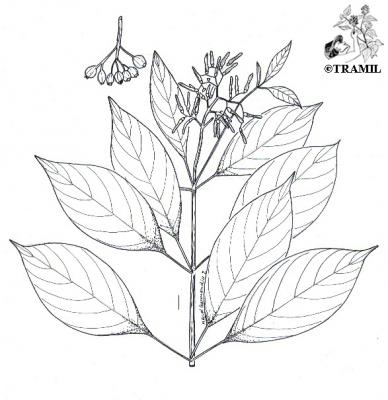
Shrub or tree 1- 6 m tall. Leaves in whorls of three, pubescent, obovate to lanceolate, acuminate, 5-15 cm x 2.5-6.5 cm; inflorescence 3-15 cm long; corolla, tubular, 1.5 to 2 cm long, orange or reddish, puberulous. Fruit subglobose or ellipsoid, 6-10 mm in diameter, red when unripe, and black when ripe.
Voucher(s)
Rouzier,161,SOE Jiménez,32,JBSD
Pharmacopoeia
Ed.2References
1 WENIGER B, ROUZIER M, 1986 Enquête TRAMIL. Service Oecuménique d'Entraide SOE, Port au Prince, Haïti.
2 WHO, 1991 Pautas para la evaluación de medicamentos herbarios WHO/TRM/91.4 (original inglés). Programa de Medicina Tradicional, OMS, Ginebra, Suiza.
3 ESPOSITO-AVELLA M, GUPTA M, 1986 Evaluación fitoquímica y farmacológica deHamelia patens yTerminalia catappa. Centro de Investigaciones Farmacognósticas de la Flora Panameña CIFLORPAN, Facultad de Farmacia, Universidad de Panamá, Panamá, Panamá.
4 ESPOSITO-AVELLA M, BROWN P, TEJEIRA I, BUITRAGO R, BARRIOS L, SANCHEZ C, GUPTA M, CEDENO J, 1985 Pharmacological screening of Panamanian medicinal plants. Part I. Int J Crude Drug Res 23(1):17-25.
5 RIPPERGER H, 1977 Isolation of isopteropodine from Hamelia patens. Pharmazie 32(H7):415-416.
6 BORGES DEL CASTILLO J, MANRESA-FERRERO MT, RODRIGUEZ-LUIS F, 1981 Oxindole alkaloids from Hamelia patens Jacq. Proc 1st Int. Conf. Chem. Biotechnol. Biol. Act Nat. Prod., Sofia, Bulgaria. Bulgarian Acad Sci Sofia 3(1):70-73.
7 CHAUDHURI PK, THAKUR RS, 1991 Hamelia patens, a new source of ephedrine. Planta Med 57(2):199.
8 ADAMS AA, LEE EF, MABRY TJ, 1989 HPLC study of oxindole alkaloids fromHamelia patens. Rev Latinoamer Quim 20(2):71-72.
9 AQUINO R, CIAVATTA L, DE SIMONE F, PIZZA C, 1990 A flavanone glycoside fromHamelia patens. Phytochemistry 29(7):2358-2360.
10 GUPTA M, ESPOSITO-AVELLA M, 1988 Evaluación química y farmacológica de algunas plantas medicinales de TRAMIL. Centro de Investigaciones Farmacognósticas de la Flora Panameña CIFLORPAN, Facultad de Farmacia, Universidad de Panamá, Panamá, Panamá.
11 MISAS C, HERNANDEZ N, ABRAHAM A, 1979 Contribution to the biological evaluation of Cuban plants. I. Rev Cub Med Trop 31(1):5-12.
12 CARBALLO A, 1995 Plantas medicinales del Escambray cubano. Apuntes científicos. Laboratorio provincial de producción de medicamentos, Sancti Spiritus, Cuba.
13 CACERES A, GIRON L, ALVARADO S, TORRES M, 1987 Screening of antimicrobial activity of plants popularly used in Guatemala for the treatment of dermatomucosal diseases. J Ethnopharmacol 20(3):223-237.
14 LOPEZ ABRAHAM A, ROJAS-HERNANDEZ N, JIMENEZ-MISAS C, 1979 Plant extracts with cytostatic properties growing in Cuba. Rev Cubana Med Trop 31(2):105-112.
15 NEGWER M, 1987 Organic chemical drugs and their synonyms (an international survey). 6th ed. Berlin, Germany: Akademie Verlag.
16 DUKE J, 1992 Handbook of biologically active phytochemicals and their bioactivities. Boca Raton, USA: CRC Press.
17 LAMAISON J, PETITJEAN-FREYTET C, CARNAT A, 1990 Rosmarinic acid, total hydroxycinnamic derivative contents and antioxidant activity of medicinal Apiaceae, Boraginaceae & Lamiceae. Ann Pharm Fr 48(2):103-108.