Vernacular names
(In territories with significant traditional TRAMIL use)
Guadeloupe:
- carapate
- karapat
- karapat blanc
Barbados:
- castor oil
Dominica:
- cawapat
Saint Lucia:
- cawapat
Dominican Republic:
- higuera
Haiti:
- maskèti
Martinique:
- palma Kristi
- ricin
Botanical description
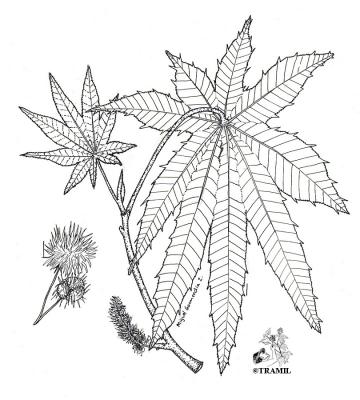
Monecious, short-lived shrub or small tree, more than 5 m high with copious clear sap. Leaves spiral, long-stalked, blades 60 cm. broad, peltate, palmately divided into 7 or more lobes; inflorescence terminal panicle to 20 cm long or more; male flowers clustered at the base of pedicels with white calyx ca, 9 mm long, female flowers at the top of pedicels with calyx deciduous; fruit a capsule oblong about 2.5 cm long, spiny prickles; seeds elliptic,10-17 mm long, mottled grey or brown.
Voucher(s)
Faujour,10,BAR
Rouzier,70,SOE
Jiménez,47,JBSD
Longuefosse & Nossin,34,HAVPMC
Pharmacopoeia
Ed.2References
1 WENIGER B, ROUZIER M, 1986 Enquête TRAMIL. Service Oecuménique d'Entraide SOE, Port au Prince, Haïti.
2 JEAN-PIERRE L, 1988 TRAMIL survey. St Lucia national herbarium, Castries, St Lucia.
3 EDOUARD JA, 1992 Enquête TRAMIL. Lycée agricole, Baie-Mahault, Guadeloupe.
4 GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984 Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
5 CHARLES C, 1988 TRAMIL survey. Movement for Cultural Awareness MCA, Roseau, Dominica.
6 WENIGER B, 1987-88 Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
7 LONGUEFOSSE JL, NOSSIN E, 1990-95 Enquête TRAMIL. Association pour la valorisation des plantes médicinales de la Caraïbe AVPMC, Fort de France, Martinique.
8 FAUJOUR A, MURREY D, CHELTENHAM-CORBIN B, CARRINGTON S, 2003 TRAMIL survey. enda-caribbean, IICA & UAG, Saint Thomas, Barbados.
9 WHO, 1991 Guidelines for the assessment of herbal medicines. WHO/TRM/91.4. Programme on Traditional Medicines, WHO, Geneva, Switzerland.
10 HEGNAUER R, 1973 Chemotaxonomy der Pflanzen. Basel, Schweiz: Birkhauser Verlag.
11 CHONKEL A, 1985 A propos de quelques graines toxiques existant à la Guadeloupe. Thèse Pharmacie, Montpellier, France.
12 DUKE JA, 1992 Handbook of phytochemical constituents of GRAS herbs and other economic plants. Boca Raton, USA: CRC Press.
13 DUKE JA, ATCHLEY AA, 1986 Handbook of proximate analysis tables of higher plants.Boca Raton, USA: CRC Press. p140.
14 DE SOUSA M, Matos ME, Matos FJ, MACHADO MI, CRAVEIRO AA,1991 Constituintes químicos ativos de plantas medicinais Brasileiras.Laboratorio de produtos naturais, Fortaleza, Brasil: Ceará Edições UFC.
15 TSUPRIENKOVA T, 1982 Patente de autor de champú para el lavado del cabello (título original en ruso). URSS, A61K 7/06(53).
16 WENIGER B, 1992 Activités biologiques (cytotoxicité, effet sur la croissance, effet immunomodulateur) de drogues végétales de la Caraïbe utilisées par voie locale contre les brûlures, dans des systèmes de cellules animales et humaines en culture. Faculté de Pharmacie, Université de Strasbourg, Illkirch, France. TRAMIL VI, Basse Terre, Guadeloupe, UAG/enda-caribe.
17 VERPOORTE R, DIHAL PP, 1987 Medicinal plants of Surinam IV. Antimicrobial activity of some medicinal plants. J Etnopharmacol 21(3):315-318.
18 MISAS CA, HERNANDEZ NM, ABRAHAM AM, 1979 Contribution to the biological evaluation of Cuban plants. I. Rev Cub Med Trop 31:5-12.
19 TANIRA MO, AGEEL AM, AL-SAID MS, 1989 A study on some Saudi medicinal plants used as diuretics in traditional medicine. Fitoterapia 60(5):443-447.
20 CECIL, RUSELL LA FAYETTE, 1987 Compendio de Medicina Interna. Madrid, España: Ed. Interamericana.
21 MARTINEZ MJ, LOPEZ M, MOREJON Z, BOUCOURT E, FUENTES V, MORON F, 2005 Irritabilidad dérmica primaria de semillas frescas peladas y machacadas de Ricinus communisL. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Ciencias Médicas “Dr. Salvador Allende”, La Habana, Cuba.
22 MARTINEZ MJ, MOREJON Z, BOUCOURT E, FUENTES V, MORON F, 2003 Irritabilidad dérmica primaria de hoja seca y de hoja fresca de Ricinus communis L. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Medicina “Dr. Salvador Allende”, Cerro, C. Habana, Cuba.
23 WEE YC, GOPALAKRISHNAKONE P, CHAN A, 1988 Poisonous plants in Singapore - a colour chart for identification with symptoms and signs of poisoning. Toxicon 26(1):47.
24 FERNANDO R, 1988 Plant poisoning in Sri Lanka. Toxicon 26(1):20.
25 CANIGUERAL S, 2003 Ricinus comunis. Vademecum de Fitoterapia, Editorial Masson, Barcelona, España, Jul.30,2003. URL: http://www.masson.es/book/fitoterapia.html
26 ALONSO J, 1998 Tratado de fitomedicina. Bases clínicas y farmacológicas. Buenos Aires, Argentina: ISIS ediciones SRL. p840.
27 KANERVA L, ESTLANDER T, JOLANKI R, 1990 Long-lasting contact urticaria from castor bean. J Amer Acad Dermatol 23(2):351-355.
28 PERIS JB, STUBING G, 2003 Ricinus comunis. Vademecum de Fitoterapia, Editorial Masson, Barcelona, España, Jul.30,2003. URL: http://www.masson.es/book/fitoterapia.html
29 BALZ E, BOYER A, BURAUD M, 2007 Enquête TRAMIL à Marie-Galante. U. Bordeaux 3, U. Paris XI Chatenay-Malabry, UAG, Guadeloupe.
30 BOYER A, BURAUD M, 2007 Enquête TRAMIL à La Désirade. U. Paris XI Chatenay-Malabry, UAG, Guadeloupe.
31 BOULOGNE Isabelle, 2008 Enquête TRAMIL à Terre-de-Haut, Les Saintes, UAG, Guadeloupe (FWI).