Vernacular names
(In territories with significant traditional TRAMIL use)
Haiti:
- medsiyen
Dominican Republic:
- piñón, piñón botija
Geographical distribution
Originally from tropical America, it has become naturalised in other tropical regions.
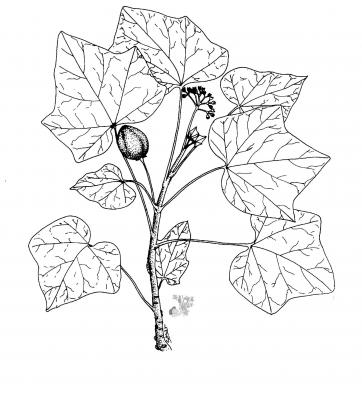
Botanical description
Shrub or small tree of up to 5 m, with milky yellow or reddish latex. Rounded-ovate leaves, 3-5 lobate, cordate at the base, sharp or acuminate at the apex. Small, dense, long-pedunculate cymes; whitish, oblong-ovate, densely pilose petals. Ellipsoid capsule, 2.5 to 4 cm; 2 cm seeds, blackish.
Voucher(s)
Rouzier,69,SOE
Pimentel,1120, JBSD
Girón,237,CFEH
Ochoa,250,HPMHV
Veloz,3039,JBSD
Pharmacopoeia
Ed.3References
- WENIGER B, ROUZIER M, 1986
Enquête TRAMIL. Service Oecuménique d’Entraide SOE, Port au Prince, Haïti.
- WENIGER B, 1987-88
Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
- LAGOS-WITTE S, 1988-89, 1996
Encuesta TRAMIL. Laboratorio de Histología Vegetal y Etnobotánica, Dep. de Biología, Universidad Nacional Autónoma de Honduras UNAH, Tegucigalpa, Honduras.
- GIRON L, 1988
Encuesta TRAMIL (Costa atlántica). Centro Mesoamericano de Tecnología CEMAT, Guatemala, Guatemala.
- CASTILLO D, RODRIGUEZ S, DE LOS SANTOS C, BELEN A, 2003
Encuesta TRAMIL, Zambrana, Cotui. Dep. de Botánica, Jardín Botánico Nacional, Santo Domingo, Rep. Dominicana.
- WENIGER B, SAVARY H, DAGUIHL R, 1984
Tri phytochimique de plantes de la liste TRAMIL. Laboratoire de chimie des substances naturelles, Faculté de Médecine et de Pharmacie, Université d’Etat d’Haïti, Port au Prince, Haïti. TRAMIL I, Port au Prince, Haïti, Fac. de Médecine/enda-caribe.
- VAN DEN BERG A, HOARSTEN S, KETTENES-VAN-DEN BOSCH J, KROES B, BEUKELMAN C, LEEFLANG B, LABADIE R, 1995
Curcacycline A - a novel cyclic octapeptide isolated from the latex of Jatropha curcas L. Febs Lett 358(3):215-218.
- NATH LK, DUTTA SK, 1991
Extraction and purification of curcain, a protease from the latex of Jatropha curcas Linn. J Pharm Pharmacol 43(2):111-114.
- HEGNAUER R, 1973
Chemotaxonomy der Pflanzen. Basel & Stuttgart: Birkhauser Verlag. 6:882.
- SUBRAMANIAN SS, NAGARAJAN S, SULOCHANA N, 1971
Flavonoids from some Euphorbiaceous plants. Phytochemistry 10:2548-2549.
- HUFFORD CD, OGUNTIMEIN BO, 1978
Non-polar constituents of Jatropha curcas. Lloydia 41:161-165.
- HUSSAIN H, DEENI Y, 1991
Plants in Kano ethomedicine; screening for antimicrobial activity and alkaloids. Int J Pharmacog 29(1):51-56.
- LE GRAND A, WONDERGEM PA, 1986
Activités antimicrobiennes et études bibliographiques de la toxicologie de dix plantes médicinales de la Caraïbe. Rapport TRAMIL. Dép. de Pharmacognosie, Universités de Groningen & Leyden, Hollande. TRAMIL II, Santo Domingo, Rep. Dominicana, UASD/enda- caribe.
- SOLIS PN, RODRIGUEZ N, ESPINOSA A, GUPTA MP, 2004
Estudio antimicrobiano de algunas plantas TRAMIL con usos en Martinica. Informe TRAMIL. Centro de Investigaciones Farmacognósticas de la Flora Panameña CIFLORPAN, Facultad de Farmacia, Universidad de Panamá, Panamá, Panamá.
- OLMEDO D, RODRIGUEZ N, ESPINOSA A, VASQUEZ Y, GUPTA MP, 2005
Ensayo antimicrobiano de algunas especies con usos significativos TRAMIL-Centroamérica. Informe TRAMIL. Centro de Investigaciones Farmacognósticas de la Flora Panameña CIFLORPAN, Facultad de Farmacia, Universidad de Panamá, Panamá, Panamá.
- GARCIA -GONZALEZ M, FERNANDEZ A. ARGUEDAS R, MONTERO S, 2005
Efecto antiinflamatorio en ratones, en aplicación tópica, del extracto acuoso (decocción) de las hojas frescas de Jatropha curcas dosis única. Informe TRAMIL. PRONAPLAMED. Depto de Fisiología, Escuela de Medicina, Universidad de Costa Rica, San Pedro, Costa Rica.
- THOMASO O, 1989
Re-examination of the antimicrobial activities of Xylopia aethiopica, Carica papaya, Ocimun gratissimum and Jatropha curcas. Fitoterapia 60(2):147-155.
- MARROQUIN E, BLANCO JA, GRANADO S, CACERES A, MORALES C, 1997
Clinical Trial of Jatropha curcas sap in the treatment of common warts. Fitoterapia 68(2):160-162.
- HERRERA J, 1990
Determinación de actividades biológicas de vegetales utilizados en medicina tradicional. Informe TRAMIL. Laboratorio de fitofarmacología, Dep. de Farmacología, Facultad de Salud, Universidad del Valle, Cali, Colombia.
- LOPEZ M, MOREJON Z, BACALLAO Y, FUENTES V, MORON F, 2009
Irritabilidad dérmica primaria de hoja fresca de Jatropha curcas L. Informe TRAMIL. Facultad de Ciencias Médicas “Dr. Salvador Allende”, Laboratorio Central de Farmacología, C. Habana, Cuba.
- WEE Y, GOPALAKRISHNAKONE P, CHAN A, 1988
Poisonous plants in Singapore - a colour chart for identification with symptoms and signs of poisoning. Toxicon 26(1):47.