Senna alata
Vernacular names
(In territories with significant traditional TRAMIL use)
Guatemala:
- barajo
Dominican Republic:
- guajabo
Martinique:
- kasyalata
Venezuela:
- majagüillo
- tarantantán
St Vincent and Grenadines:
- river grava
- river senna
Trinidad and Tobago:
- wild guava
- wild senna
Botanical description
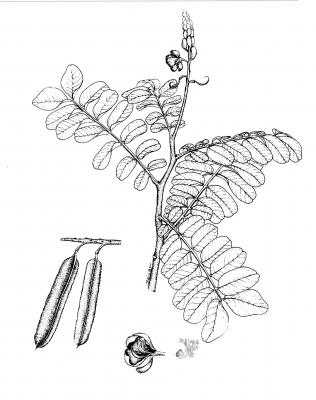
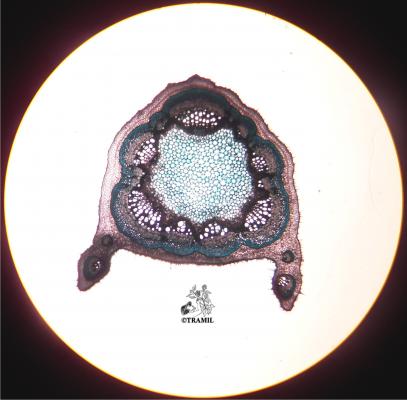
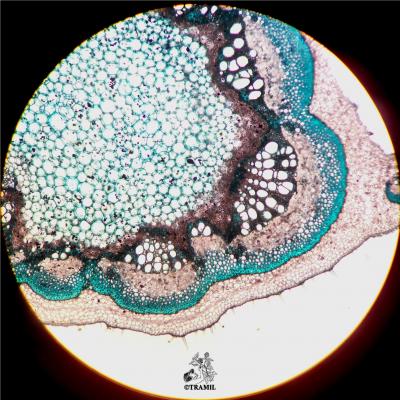
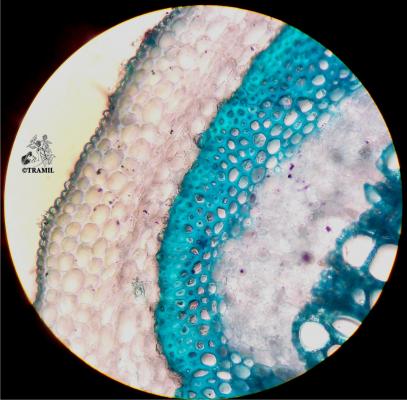
A short-lived shrub up to 3 m high. Leaves alternate, compound pinnate, with 6-12 pairs of broadly oblong leaflets, blunt at the tip, unequal at the base, the terminal pair about 15 x 8 cm; inflorescence an axillary, compact raceme ca. 28 cm long; flowers golden yellow, subtended by a yellow-orange bract, 2.2 -2.5 cm long; pod 4-winged, 11-19 x 2-2.3 cm, black when ripe; seeds ca. 6 mm long, brown, angular, arranged transversely in the pod.
Voucher(s)
Delens,138,VEN
Longuefosse &Nossin,36,HAVPMC
Girón,154,CFEH
Jiménez,3528,JBSD
Balland,44,HVB
Delaigue,25,NHTT
Pharmacopoeia
Ed.2References
1 DELENS M, 1990-92 Encuesta TRAMIL. Centro al Servicio de la Acción Popular CESAP, Caracas, Venezuela.
2 LONGUEFOSSE JL, NOSSIN E, 1990-95 Enquête TRAMIL. Asociation pour la valorisation des plantes médicinales de la Caraïbe AVPMC, Fort de France, Martinique.
3 GIRON L, 1988 Encuesta TRAMIL (Costa atlántica). Centro Mesoamericano de Tecnología CEMAT, Guatemala, Guatemala.
4 Castillo D, Rodriguez S, de los Santos C, Belen A, 2003 Encuesta TRAMIL (región Este). Dep. de Botánica, Jardín Botánico Nacional, Santo Domingo, República Dominicana.
5 BALLAND V, GLASGOW A, SPRINGER F, GAYMES G, 2004 TRAMIL survey. enda-caribbean, IICA, UAG & U.PARIS XI, Saint Vincent.
6 HARRISON J, GARRO CV, 1977 Study on anthraquinone derivatives fromCassia alata L. (Leguminosae). Rev Peru Bioquim 1(1):31-33.
7 MULCHANDANI NB, HASSARAJANI SA, 1975 Isolation of 1,3,8-trihydroxy-2-methylanthraquinone fromCassia alata (leaves). Phytochemistry 14:2728b.
8 HAUPTMANN H, NAZARIO LL, 1950 Some constituents of the leaves of Cassia alata. J Am Chem Soc 72:1492-1495.
9 RAO CK, SUBHASHINI G, 1986 Saponins & leucoanthocyanins in Cassia L. Curr Sci 55(6):320-321.
10 SMOLENSKI SJ, SILINIS H, FARNSWORTH NR, 1975 Alkaloid screening. VI. Lloydia 38(3):225-255.
11 RAI MK, UPADHYAY S, 1988 Screening of medicinal plants of Chindwara district against Trichophyton mentagrophytes: a causal organism of Tinea pedis. Hindustan Antibiot Bull 30(1/2):33-36.
12 FIALLO M, VAZQUEZ TINEO M, 1992 Evaluación in vitro de plantas usadas en afecciones de la piel: Extractos vegetales antimicóticos y antimicrobianos. Informe TRAMIL. CIBIMA, Fac de Ciencias, Universidad Autónoma UASD, Santo Domingo, Rep. Dominicana.
13 ACHARARIT C, PANYAYONG W, RUCHATAKOMUT E, 1983 Inhibitory action of some Thai herbians (medicinal plants) to fungi. Mahidol Univ Fac Pharm Bangkok, Thailand.
14 BENJAMIN TV, LAMIKANRA A, 1981 Investigation ofCassia alata, a plant used in Nigeria in the treatment of skin diseases. Quart J Crude Drug Res 19(2/3):93-96.
15 CACERES A, LOPEZ BR, GIRON MA, LOGEMANN H, 1991 Plants used in Guatemala for the treatment of dermatophytic infections. 1. Screening for antimycotic activity of 44 plant extracts. J Ethnopharmacol 31(3):263-276.
16 FUZELLIER MC, MORTIER F, LECTARD P, 1982 Activité antifongique deCassia alata L. Ann Pharm Fr 40(4):357-363.
17 MATTA DC, 2000 Determinación de la actividad anti Neisseria gonorrhoeae de extractos vegetales por un método de dilución en agar (Tesis de química-biología). Facultad de Ciencias Químicas y Farmacia, Universidad de San Carlos USAC, Guatemala, Guatemala.
18 PALANICHAMY S, AMALA BHASKAR E, BAKTHAVATHSALAM R, NAGARAJAN S, 1991 Wound healing activity of Cassia alata. Fitoterapia 62(1):153-156.
19 DAMODARAN S, VENKATARAMAN S, 1994 A study on the therapeutic efficacy of Cassia alata, Linn. Leaf extract against pityriasis versicolor. J Ethnopharmacol 42(1):19-23.
20 MOKKHASMIT M, NGARMWATHANA W, SAWASDIMONGKOL K, PERMPHIPHAT U, 1971 Pharmacological evaluation of Thai medicinal plants (cont.). J Med Assoc Thai 54(7):490-504.
21 MARTINEZ MJ, MOREJON Z, LOPEZ M, BOUCOURT E, BARCELO H, LAYNEZ A, FUENTES V, MORON F, 2003 Clases Toxicidad Aguda (CTA) de hoja frescade Senna alata (L.) Roxb. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Ciencias Médicas “Dr. Salvador Allende”, La Habana, Cuba.
22 LOPEZ M, MARTINEZ MJ, MOREJON Z, BOUCOURT E, FERRADA C, FUENTES V, MORON F, 2005 Irritabilidad dérmica primaria de la maceración acuosa de hoja fresca de Senna alata (L.) Roxb. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Medicina “Dr. Salvador Allende”, Cerro, C. Habana, Cuba.
23 MOKKHASMIT M, SWATDIMONGKOL K, SATRAWAHA P, 1971 Study on toxicity of Thai medicinal plants. Bull Dept Med Sci 12(2-4):36-65.
24 VIZOSO A, RAMOS A, VILLAESCUSA A, BETANCOURT J, GARCIA A, PILOTO J, DECALO M, 2002 Passiflora incarnataL. y Senna alata (L.) Roxo: Estudio toxicogenético que emplea 2 sistemas de ensayos a corto plazo. Rev Cubana Plant Med 7(1):27-31.
25 DELAIGUE J, 2005 TRAMIL survey. PRDI, Tobago House of Assembly, Scarborough, Tobago.
26 LOGARTO PARRA A, SILVA YHEBRA R, GUERRA SARDINAS I, IGLESIAS BUELA L, 2001 Comparative study of the assay of Artemia salina L. and the estimate of the medium lethal dose (LD50 value) in mice, to determine oral acute toxicity of plant extracts. Phytomedicine 8(5):395-400.
27 MARTINEZ MJ, BETANCOURT J, LOPEZ M, MOREJON Z, FUENTES V, MORON F, 2005 Clases tóxicas agudas tópica de hoja fresca machacada de Senna alata. Informe TRAMIL. Laboratorio Central de Farmacología, Facultad de Ciencias Médicas “Dr. Salvador Allende”, La Habana, Cuba.