Vernacular names
(In territories with significant traditional TRAMIL use)
Colombia:
- papa
Costa Rica:
- papa
Guatemala:
- papa
Botanical description
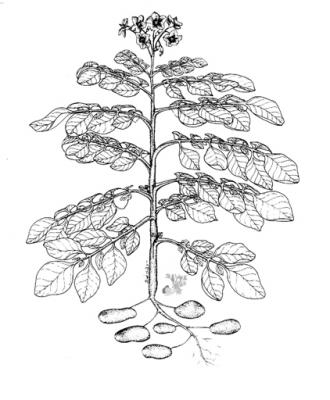
Herbaceous branched annual 0.3-1m high, with a mass of fine fiberous adventitious roots and swollen stem tubers. Leaves pinnate with small interjected leaflets between the main pinnae; flowers white, red or purple; fruit a small inedible berry.
Voucher(s)
Girón,279,CFEH
Pharmacopoeia
Ed.2References
1 RESTREPO O, 1988 Encuesta TRAMIL. Suna Hisca, Bogotá, Colombia.
2 GIRON L, 1988 Encuesta TRAMIL (Costa atlántica). Centro Mesoamericano de Tecnología CEMAT, Guatemala, Guatemala.
3 OCAMPO R, 1988 Encuesta TRAMIL (Costa atlántica), Instituto de Desarrollo Agrario, Universidad de Costa Rica, San José, Costa Rica.
4 WHO, 1991 Pautas para la evaluación de medicamentos herbarios WHO/TRM/91.4 (original inglés). Programa de Medicina Tradicional, OMS, Ginebra, Suiza.
5 CALLAGHAN JM, TOH BH, PETTITT JM, HUMPHRIS DC, GLEESON PA, 1990 Poly-N-acetyllactosamine-specific tomato lectin interacts with gastric parietal cells. Identification of a tomato lectin binding 60-90-X-10(3) Mr-membrane glycoprotein of tubulovesicles. J Cell Sci 95(4):563-576.
6 DE STEFANO JA, CUSHION MT, TRINKLE LS, WALZER PD, 1989 Lectins as probes to Pneumocystis carinii surface glycocomplexes. J Protozool 36(1):65-66S.
7 WILLAMAN J, HUI-LIN-LI, 1970 Alkaloid-bearing plants and their contained alkaloids. 1957-1968. Lloydia 33(3A)Supp.
8 ANDERSEN OM, OPHEIM S, AKSNES DW, FROYSTEIN NA, 1991 Structure of petanin, an acylated anthocyanin isolated from Solanum tuberosum, using homonuclear and heteronuclear two-dimensional nuclear magnetic resonance techniques. Phytochem Anal 2(5):230-236.
9 MONNET R, SABON F, VIALA A, 1956 Pommes de terre à teneur élevée en solanine. Food Sci Abs 28(3):314.
10 LIOGIER A, 1990 Plantas medicinales de Puerto Rico y del Caribe. San Juan, Puerto Rico: Iberoamericana de Ediciones, Inc.
11 PARIS R, MOYSE H, 1981 Précis de Matière Médicale. Paris, France: Ed. Maloine.
12 DUKE JA, ATCHLEY AA, 1986 Handbook of proximate analysis tables of higher plants. Boca Raton, USA: CRC Press.
13 SANTOS AE, COUSIN LA, 1989 Efecto de los extractos acuosos de algunas plantas medicinales de Honduras en la prevención de úlceras gástricas en ratas (Tesis de medicina). Unidad de Farmacología, Facultad de Ciencias Médicas, Universidad Nacional Autónoma de Honduras UNAH, Tegucigalpa, Honduras.
14 HOJIMA Y, PISANO JJ, COCHRNE CG, 1983 Survey of plant inhibitors of polymorphonuclear leukocyte elastase, pancreatic elastase, cathepsin G, cathepsin B, Hageman-factor fragments, and other serine proteinases. Biochem Pharmacol 32(6):985-990.
15 MIWA M, KONG ZL, SHINOHARA K, WATANABE M, 1990 Macrophage stimulating activity of foods. Agr Biol Chem 54(7):1863-1866.
16 QUIRCE S, DIEZ GOMEZ ML, HINOJOSA M, CUEVAS M, URENA V, RIVAS MF, PUYANA J, CUESTA J, LOSADA E, 1989 Housewives with raw potato-induced bronchial asthma. Allergy 44(8):532-536.
17 KIM TH, KIM SH, LEE YS, CHOI SY, YOO SY, JANG JJ, 1994 Protective effects of potato extracts and 16,16-dimethyl prostaglandin E2 on the induction of hepatic foci by cotreatment of gamma radiation and diethylnitrosamine. Anticancer Res 14(5a):1979-1982.
18 BAKER D, KEELER R, GAFFIELD W, 1987 Lesions of potato sprout and extracted potato sprout alkaloid toxicity in Syrian hamsters. J Toxicol Clin Toxicol 25(3):199-208.
19 CANIGUERAL S, VILA R, RISCO E, PEREZ F, PORTILLO A, FREIXA B, MILO B, VANACLOCHA B, RIOS JL, MORALES MA, ALONSO JR, BACHILLER LI, PERIS JB, STUBING G, 2002 Solanum tuberosum. Vademecum de Fitoterapia, Editorial Masson, Barcelona, España, Nov.20,2003. URL: http://www.masson.es/book/fitoterapia.html
20 PAZOS L, COTO T, CAIZA F, 2009
Irritación dérmica, piel sana en conejos, tubérculo fresco rayado de Solanum tuberosum. Informe TRAMIL. Laboratorio de Ensayos Biológicos, LEBi, Universidad de Costa Rica, San Pedro, Costa Rica.