Vernacular names
(In territories with significant traditional TRAMIL use)
Dominican Republic:
- cacao
Botanical description
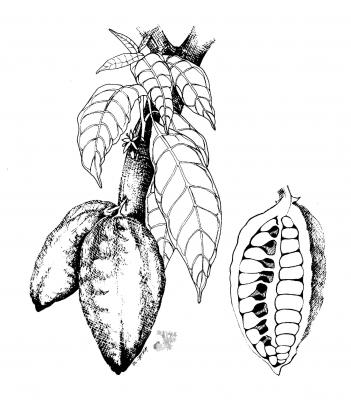
Small tree, 6- 8 (-12-14) m high. Leaf, alternate, simple, entire, sinuate, petiole 1-2 cm, blades obovate-elliptic to oblong elliptic, 16-40 x 4.5-13 cm, base rounded to obtuse, apex acute; inflorescence axillary, 1-many flowered, clustered cymes, cauliflorous; petals white 4-4.5 mm long; fruit a glabrous, ellipsoid, leathery berry, 10-20 x 8-10 cm wide, ripening green, yellow, red or purple; seeds brown, ovoid or elliptic, 2-4x1.2-2 cm long with a sweet surrounding pulp.
Voucher(s)
Girón,281,CFEH
Pharmacopoeia
Ed.2References
1 GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984 Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
2 KENYHERCZ TM, KISSINGER PT, 1978 Determination of selected acidic, neutral, and basic natural products in cacao beans and processed cocoa. Liquid chromatography with electrochemical detection. Lloydia 41(2):130-139.
3 SJOBERG AM, RAJAMA J, 1984 Simple method for the determination of alkaloids in cocoa using paper chromatography and UV spectrometry. J Chromatogr 295(1):291-294.
4 GURNEY KA, EVANS LV, ROBINSON DS, 1991 Extraction of purine alkaloids from cocoa tissues and determination by high-performance liquid chromatography. Phytochem Anal 2(1):15-19.
5 RIGGIN RM, KISSENGER PT, 1976 Identification of salsolinol as a phenolic component in powdered cocoa and cocoa-based products. J Agric Food Chem 24(4):900.
6 VITZTHUM OG, WERKHOFF P, HUBERT P, 1975 Volatile components of roasted cocoa: basic fraction. J Food Sci 40:911-916.
7 PORTER LJ, MA Z, CHAN BG, 1991 Cacao procyanidins: major flavanoids and identification of some minor metabolites. Phytochemistry 30(5):1657-1663.
8 THOMPSON RS, JACQUES D, HASLAM E, TANNER RJN, 1972 Plant proanthocyanidins. Part I. Introduction: the isolation, structure, and distribution in nature of plant procyanidins. J Chem Soc Perkin Trans 1:1387-1399.
9 VAN PRAAG M, STEIN HS, TIBBETTS MS, 1968 Steam volatile aroma constituents of roasted cocoa beans. J Agric Food Chem 16:1005.
10 LIMA VASCONCELOS MN, LEAO DE SILVA M, SOARES MAIA JG, GOTTLIEB OR, 1975 Chemical study of cupu seeds. Acta Amazonica 5:293.
11 ADOMAKO D, 1977 Fatty acid composition and characteristics of Pentadesma butyracea fat extracted from Ghana seeds. J Sci Food Agr 28:384-386.
12 DUKE JA, ATCHLEY AA, 1986 Handbook of proximate analysis tables of higher plants. Boca Raton, USA: CRC Press, p159.
13 PEREZ C, ANESINI C, 1994 Antibacterial activity of alimentary plants against Staphylococcus aureus growth. Amer J Chinese Med 22(2):169-174.
14 MORRISSEY RB, BURKHOLDER BD, TARKA SM, 1984 Effects of cocoa upon the growth of weanling male Sprague-Dawley rats fed fluid whole milk diets. Nutr Rep Int 29(2):263-271.
15 OSAKABE N, YAMAGISHI M, SANPPONGI C, TAKIZAWA T, ADACHI T 1995 Antioxidant-containing beverages for preventing gastric ulcer. Patent-Japan Kokai Tokkyo Koho-07 274,894.
16 HARDMAN JG, GILMAN AG, LIMBIRD LE (Eds.), 1996 The pharmacological basis of therapeutics. 9th ed. New York, USA: McGraw-Hill Professional Publishing, International Edition.
17 KATZUNG BG (Ed.), 1999 Basic and clinical pharmacology. McGraw-Hill Education, New York, USA.
18 PDR Herbals, 2003 Cocoa: Theobroma cacao. The PDR for Herbal Medicines, PDR Electronic Library, Medical Economics Company, PhytoPharm US Institute for Phytopharmaceuticals, Metuchen, USA, Oct. 8, 2003. URL: http://www.mdcc.edu/medical/library
19 ANON (Select Committee on GRAS Substances), 1976 GRAS status of foods and food additives. Washington DC,USA: Food and Drug Administration, Department of Health and Human Services, Office of the Federal Register National Archives and Records Administration41, 38644
20 FLORIPE A, ALTAMIRANO V, 1998 Plantas que curan. Managua, Nicaragua: Imprimatur. p35.