Vernacular names
(In territories with significant traditional TRAMIL use)
Dominican Republic:
- malagueta
Geographical distribution
Southeast Mexico, Central America and the Antilles
Botanical description
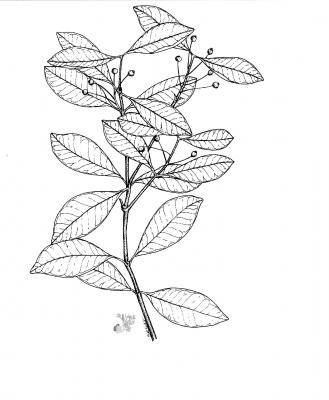
Tree of up to 20 m; smooth and straight trunk. Opposite, coriaceous, ovate or elliptical leaves of up to 20 cm long, very aromatic. Panicles of about 12 cm in length with many flowers; flowers mostly grouped at the tip of the inflorescence, sessile; calyx with 4 rounded lobes, puberulous; white suborbicular petals; numerous white stamens. Aromatic fruit, generally subglobose, verrucose, with two suborbicular seeds.
Voucher(s)
Jiménez,1503,JBSD
Pharmacopoeia
Ed.3References
1 GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984
Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
2 DUKE JA, 1988
Handbook of medicinal herbs. Boca Raton, USA: CRC.
3 KIUCHI F, HIOKI M, NAKAMURA N, MIYASHITA N, TSUDA Y, KONDO K, 1989
Screening of crude drugs used in Sri Lanka for nematocidal activity on the larva of Toxocaria canis. Shoyakugaku Zasshi 43(4):228-293.
4 TUCKER A, MACIARELLO M, LETRUM L, 1991
Volatile leaf oils of Caribbean Myrtaceae. II. Pimenta dioica (L.) Merr. of Jamaica. J Essent Oil Res 3(3):195-196.
5 PADMAKUMARI KP, SASIDHARAN I, SREEKUMAR MM, 2011
Composition and antioxidant activity of essential oil of pimento (Pimenta dioica (L) Merr.) from Jamaica. Nat Prod Res 25(2):152-160.
6 KIKUZAKI H, MIYAJIMA Y, NAKATANI N, 2008
Phenolic glycosides from berries of Pimenta dioica. J Nat Prod 71(5):861-865.
7 KIKUZAKI H, SATO A, MAYAHARA Y, NAKATANI N, 2000
Galloylglucosides from berries of Pimenta dioica. J Nat Prod 63(6):749-752.
8 YOSHIMURA M, AMAKURA Y, YOSHIDA T, 2011
Polyphenolic compounds in clove and pimento and their antioxidative activities. Biosci Biotechnol Biochem 75(11):2207-2212.
9 HERRERA J, 1988
Determinación de actividades biológicas de vegetales utilizados en medicina tradicional. Informe TRAMIL. Dep. de Farmacología, Facultad de Salud, Universidad del Valle, Cali, Colombia.
10 HERRERA J, 1992
Determinación de parámetros farmacológicos usados en medicina tradicional popular en la Cuenca del Caribe. Informe TRAMIL. Dep. de Farmacología, Facultad de Salud, Universidad del Valle, Cali, Colombia.