Pouteria sapota
Vernacular names
(In territories with significant traditional TRAMIL use)
Dominican Republic:
- sapote
Botanical description
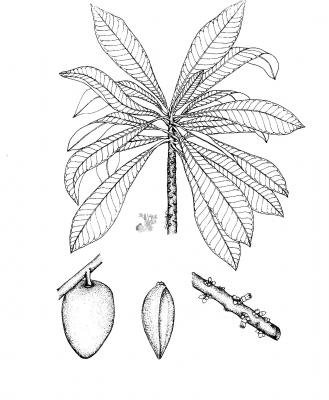
Tree, up to 30 m high, bark fissured, with milky white latex. Leaves, simple, alternate, blades oblanceolate, 10-60 x 4.5-5 cm, apex acuminate, base cuneate, glabrous above, pubescent beneath; inflorescence in clusters at defoliated nodes or cauliflorus; flowers white with corolla 9-10 mm long; fruit ellipsoid, ovoid or subglobose 8-20 cm long, brown, rough; seeds solitary, broadly obovate, deep brown to black and shiny.
Voucher(s)
Jiménez,20,JBSD
Pharmacopoeia
Ed.2References
1 GERMOSEN-ROBINEAU L, GERONIMO M, AMPARO C, 1984 Encuesta TRAMIL. enda-caribe, Santo Domingo, Rep. Dominicana.
2 JIU J, 1966 A survey of some medicinal plants of Mexico for selected biological activities. Lloydia29:250-259.
3 MISAS CAJ, HERNANDEZ NMR, ABRAHAM AML, 1979 Contribution of the biological evaluation of Cuban plants. VI. Rev Cub Med Trop 31:45-51.
4 ROJAS HERNANDEZ NM, JIMENEZ MISAS CA, LOPEZ ABRAHAM AM, HERNANDEZ SUAREZ C, 1981 Study of the inhibitory activity of plant extracts on microbial growth. Part V. Rev Cubana Farm 15:139-145.
5 LOPEZ ABRAHAM AN, ROJAS HERNANDEZ NM, JIMENEZ MISAS CA, 1981 Potential antineoplastic activity of Cuban plants. IV. Rev Cubana Farm 15(1):71-77
6 DUKE JM, 1992 Handbook of biologically active phytochemicals and their bioactivities. Boca Raton, USA: CRC Press.
7 POULTON J, KEELER R, TU T (Eds.), 1983 Handbook of natural toxins 1. New York, USA: Marcel Dekker.
8 NAHRSTEDT A, 1987 Recent developments in chemistry, distribution and biology of the cyanogenic glycosides. In: HOSTETTMAN K, LEA, JP (Eds.), Biologically Active Natural Products. Oxford, USA: Oxford Science Publications. pp167-184,213-234.
9 KLAASSEN C, AMDUR D, MARY O, 1986 Toxicology, the basic science of poisons. 3th ed. New York, USA: McMillan Publishing Co.
Warnings
According to published and other information:
Use of the seed orally is classified as tOxic (TOX). Given the toxicity of the seed of this plant, its use is discouraged, regardless of how recognized its alleged therapeutic properties may be.
In the event of poisoning from ingestion, seek medical attention.