Cissampelos pareira
Vernacular names
(In territories with significant traditional TRAMIL use)
Haiti:
- pat chwal
Geographical distribution
Widely distributed in the tropical regions of the world.
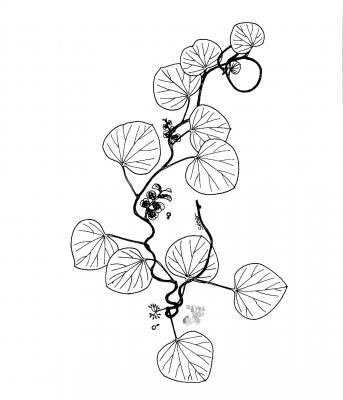
Botanical description
Herbaceous climbing plant, pubescent to tomentose. Suborbicular or ovate leaves, 3.5 to 10 cm, rounded to emarginated, often peltate. Staminate inflorescence in corymbs up to 7 cm born in the axils of the leaves; green female flowers in racemes of up to 8 cm. Subglobose drupe (fruit), reddish from 3.5 to 4.5 mm in diameter.
Voucher(s)
Voltaire,256,SOE
Pharmacopoeia
Ed.3References
1 WENIGER B, ROUZIER M, 1986
Enquête TRAMIL. Service Oecuménique d’Entraide SOE, Port au Prince, Haïti.
2 KUPCHAN SM, PATEL AC, FUJITA E, 1965
Tumor inhibitors VI. Cissampareine, new cytotoxic alkaloid from Cissampelos pareira, cytotoxicity of benzylisoquinoline alkaloids. J Pharm Sci 54:580.
3 SINGH A, DUGGAL S, SINGH J, KATEKHAY S, 2010
An inside preview of ethnopharmacology of Cissampelos pareira Linn. International J of Biological Technology 1(1):114-120.
4 BHATNAGAR AK, BHATTACHARJI S, POPLI SP, 1968
Nuclear magnetic resonance spectrum of cycleanine. Indian J Chem 6:125.
5 ANWER F, POPLI SB, SRIVASTAVA RM, KHARE MP, 1968
Studies in medicinal plants. Part III. Protoberberine alkaloids from the roots of Cissampelos pareira. Experientia 24:999.
6 RAMIREZ I, CARABOT A, MELENDEZ P, CARMONA J, JIMENEZ M, PATEL AV, CRABB TA, BLUNDEN G, CARY PD, CROFT SL, COSTA M, 2003 Cissampeloflavone, a chalcone-flavone dimer from Cissampelos pareira. Phytoche- mistry 64(2):645–647.
7 MOJAB F, KAMALINEJAD M, GHADERI N, VAHIDIPOUR HR, 2003
Screening of some species of Iranian plants. Iranian J of Pharmaceutical Research: 77-82.
8 HERRERA J, 1994
Determinación de actividades biológicas de vegetales utilizados en medicina tradicional. Informe TRAMIL. Laboratorio de Fitofarmacología, Departamento de Farmacología, Facultad de Salud, Universidad del Valle, Cali, Colombia.
9 GEORGE M, PETALAI K, 1949
Investigations on plant antibiotics. Part IV. Further search for antibiotic substances in Indian medicinal plants. Indian J Med Res 37:169-181.
10 FENG PC, HAYNES LJ, MAGNUS KE, PLIMMER JR, SHERRAT HSA, 1962
Phamacological screening of some West Indian medicinal plants. J Pharm Pharmacol 14:556-561.
11 SARAVIA A, 1992
Toxicidad de Cissampelos pareira. Informe TRAMIL. Universidad de San Carlos, Guatemala, Guatemala. TRAMIL VI, Basse Terre, Guadeloupe, UAG/enda-caribe.
12 CHAPUIS J, SORDAT B, HOSTETTMANN K, 1988
Screening for cytotoxic activity of plants used in traditional medicine. J
Ethnopharmacol 23(2/3):273-284.
13 ROY P, 1952
A preliminary note on the pharmacological action of the total alkaloids isolated from Cissampelos pareira. Indian J Med Res 40:95.